Tumor significance was defined by systematic biopsies findings: presence of Gleason grade 4 and/or maximum cancer core length on any core greater than half the total length of the core. About half of the patients in the cohort were biopsy naive and the median follow-up was 3 years. Significant cancer was detected in 5% of cases at the end of the follow-up. The other half had a previous negative biopsy and the median follow-up was 5 years. Significant cancer was detected in 4% of cases. Overall, 60 significant tumors were detected (4.8%) after 7 years of surveillance. Gleason score was 3+3, 3+4, 4+3 and >4+3 in 12, 41, 30 and 17% of cases respectively. The predominant predicting factor of the presence of a significant non-visible cancer was a PSA density >0.15, multiplying by about 7 the risk of finding a positive biopsy with a Gleason grade >3. The identified reasons for MRI false negative cases were the presence of a benign hyperplasia, prostatitis in the peripheral zone (PZ), small (<0.5cc) tumors originating in the anterior horn of the PZ, mucinous and cribriform adenocarcinoma in 32, 14, 31, 2, and 22% of cases, respectively.
This article follows the PROMIS study (2). The PROMIS study (2) demonstrated that a normal MRI has a high negative predictive value (NPV) to predict the absence of significant cancer, ranging from 72 to 89% depending on the definition of significant cancer. The rate of 89% corresponded to cancers with a Gleason score >3+4 and/or a maximum cancer core length on any core>5mm. In the article by Panebianco et al, it is surprising that cribriform cancers represented 22% of false MRI negatives, as they most often have a score of 4 or 5 on MRI (3, 4). The article also refers to the variability of NPV as a function of cancer prevalence.The higher the risk of cancer in the study population, the lower the NPV can be, without modification of the sensitivity (5). This statistical principle can therefore theoretically be applied to the NPV of a normal prostate MRI, and risk calculators have been proposed to better evaluate the cancer probability in the “pre-test” assessment of the patient by clinical and biological parameters (6), with a predominant importance, in the article by Panebianco et al. (1) of PSA density >0.15. That being said, one can wonder what is the significance of a nonvisible cancer on MRI. Based on correlations with radical prostatectomy specimens, tumors detected in areas with no MRI abnormality are, in the majority of cases, low risk Gleason score 3+4 cancers, with only a small percentage of Gleason 4 grade (7). These tumors have been considered eligible for active surveillance (8) and diagnosis of these tumors would thus not have a significant impact on the immediate management of the patient. These data are somewhat contradictory with the percentage of cancers with a Gleason score of >3+4 (47%) detected in the study by Panebianco et al (1).
Nevertheless, the article by Panebianco et al (1) illustrates how a non suspicious MRI and a biological suspicion of prostate cancer are increasingly being managed, by deferring the biopsy and only performing it if signs of progression appear (PSA level and/or increase of PIRADS score repeat MRI).
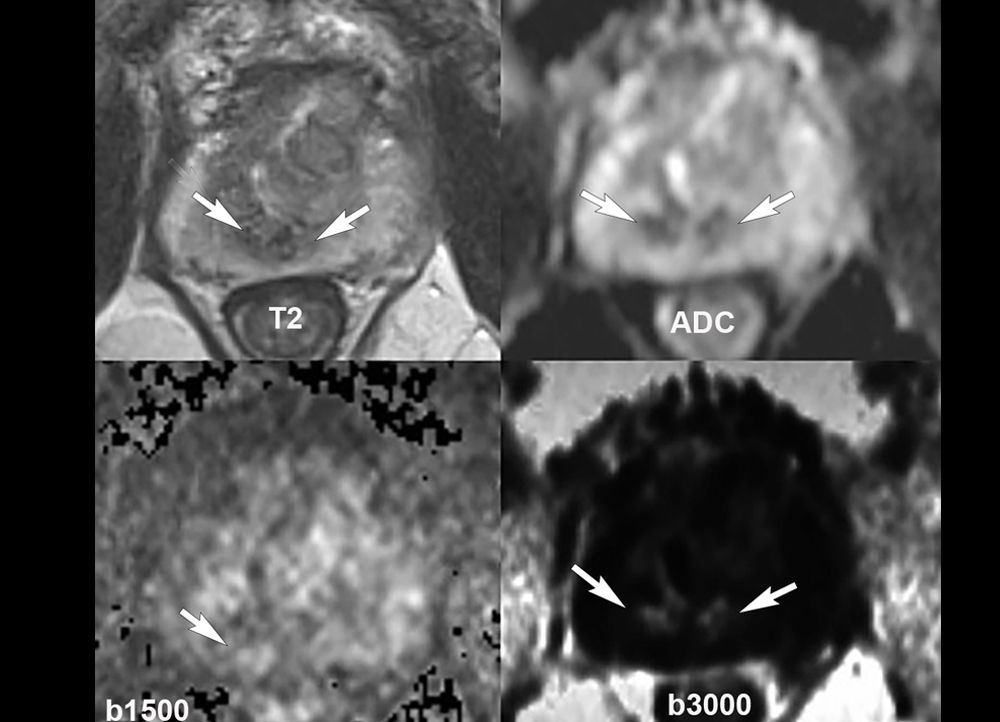
Caption Figure 1 : Appearance of a non-suspicious MRI in a patient with a PSA density <0.15. No focal restricted diffusion in the peripheral zone and no suspicious anomaly in the transition zone on the T2-weighted sequence. Arrows show the T2W hyposignal and the physiological restricted diffusion of the central zone around the ejaculatory ducts.
References :
- Panebianco V, Barchetti G, Simone G, Del Monte M, Ciardi A, Grompone MD, et al. Negative Multiparametric Magnetic Resonance Imaging for Prostate Cancer: What’s Next? Eur Urol. 2018.
- Ahmed HU, El-Shater Bosaily A, Brown LC, Gabe R, Kaplan R, Parmar MK, et al. Diagnostic accuracy of multi-parametric MRI and TRUS biopsy in prostate cancer (PROMIS): a paired validating confirmatory study. Lancet. 2017;389(10071):815-22.
- Prendeville S, Gertner M, Maganti M, Pintilie M, Perlis N, Toi A, et al. Role of Magnetic Resonance Imaging Targeted Biopsy in Detection of Prostate Cancer Harboring Adverse Pathological Features of Intraductal Carcinoma and Invasive Cribriform Carcinoma. J Urol. 2018;200(1):104-13.
- Truong M, Feng C, Hollenberg G, Weinberg E, Messing EM, Miyamoto H, et al. A Comprehensive Analysis of Cribriform Morphology on Magnetic Resonance Imaging/Ultrasound Fusion Biopsy Correlated with Radical Prostatectomy Specimens. J Urol. 2018;199(1):106-13.
- Rouviere O, Souchon R, Melodelima C. Pitfalls in interpreting positive and negative predictive values: Application to prostate multiparametric magnetic resonance imaging. Diagn Interv Imaging. 2018.
- Mehralivand S, Shih JH, Rais-Bahrami S, Oto A, Bednarova S, Nix JW, et al. A Magnetic Resonance Imaging-Based Prediction Model for Prostate Biopsy Risk Stratification. JAMA Oncol. 2018.
- Lanz C, Cornud F, Beuvon F, Lefevre A, Legmann P, Zerbib M, et al. Gleason Score Determination with Transrectal Ultrasound-Magnetic Resonance Imaging Fusion Guided Prostate Biopsies-Are We Gaining in Accuracy? J Urol. 2016;195(1):88-93.
- Garisto JD, Klotz L. Active Surveillance for Prostate Cancer: How to Do It Right. Oncology (Williston Park). 2017;31(5):333-40, 45.